By Greg F. Mathis, PhD
Brett Lumpkins, PhD
Southern Poultry Feed & Research, Inc, Athens, Georgia
Birth of poultry anticoccidials
Chemotherapy of coccidiosis in poultry was revolutionized by Levine in 1941 with the discovery of sulfonamides,1 which were active against the chicken coccidia Eimeria tenella. This discovery led to the development of many anticoccidial drugs that helped bring coccidiosis under control.
A 2014 review paper by Chapman highlighted one of the major landmark discoveries in coccidiosis control: that coccidiosis could be better managed and controlled by the continuous feeding of a low concentration of certain anticoccidial drugs.2,3 Reid stated that the availability of economical and effective anticoccidial drugs and the prophylactic feeding of these drugs supported the rapid rise of an intensive confined rearing of poultry.4
In The Biology of the Coccidia, McDougald stated that prevention programs for coccidiosis are preferable to treatment.5 The onset of coccidiosis can occur quickly, with the acute phase (morbidity and mortality) occurring before therapeutic measures can be introduced.
Most anticoccidial drugs work against multiple coccidiosis stages. This includes the early (asexual) stages that result in major damage to the intestinal tract and thus impair the health of the production bird, i.e., broiler, layer or turkey. However, therapeutic anticoccidials are limited in number, some with toxicity issues, insufficient species spectrum of activity or both.
McDougald also stated that “the logistics of timely therapeutic action, limited safe and useful choices and the structure of the poultry industry negate the usefulness of treatment as an economical or practical means of coccidiosis control in lieu of preventative medication.”6 Notwithstanding withdrawal times, continuous prophylactic feeding of anticoccidial drugs is needed for maximum protection.
Anticoccidials on the market
In 1981, there were 27 FDA-approved anticoccidial drugs in the US.7 Today, there are only 12 FDA-approved drugs for chicken coccidiosis control. The first anticoccidial drugs were synthetic. Generally, these compounds have a strong, broad-species spectrum of anticoccidial activity. The downside to the strong anticoccidial activity is that some degree of resistance/decreased sensitivity has developed to all of these products.8 There are only 7 feed-additive synthetic drugs that remain (Table 1).
Table 1: FDA-approved synthetic anticoccidial drugs and when each was introduced to the US market
| Drug | Introduced to market |
| Nicarbazine | 1956 |
| Clopidol | 1968 |
| Zoalene | 1960 |
| Amprolium | 1960 |
| Decoquinate | 1970s |
| Robenidine | 1970s |
| Diclazuril | 1991 |
Nicarbazine, one of the oldest anticoccidial drugs, was approved in 1956. There is limited documented resistance to this compound.9 Clopidol was approved in 1968, and zoalene in 1960, with both used prophylactically at 0.125% in the feed. Both were later removed from the market for an extended period.
After many years of non-use of these two drugs, anticoccidial-sensitivity tests were conducted, and it was determined that numerous coccidia field strains were sensitive to both drugs. Based on this information and the resolution of manufacturing issues, zoalene and clopidol were successfully and effectively reintroduced to the broiler market.
Another drug introduced in the 1960s was amprolium, which has its greatest activity against E. tenella and E. necatrix. Due to its high solubility, amprolium is often used as a water-administered therapeutic agent.
Decoquinate and robenidine were approved in the early 1970s and used prophylactically at 0.033% in the feed. Robenidine was first approved at 66 parts per million (ppm), with the hope that this strong activity would eliminate any chance for resistance development. Unfortunately, this assumption proved to be incorrect. Research has shown that there is a generally stronger selection for resistance with higher drug doses. The dose was reduced to 33 ppm with this new understanding of resistance.
Polyether ionophorous antibiotic anticoccidials
With the introduction of the ionophore monensin, in 1971, this class of anticoccidials became the principal product used, quickly reaching 80% to 90% of the market.
The polyether ionophorous antibiotics — ionophores — are produced by the fermentation of Streptomyces spp. or Actinomadura spp. The ionophores are placed into three groups: monovalent (monensin, narasin and salinomycin), monovalent glycosidic (maduramicin and semduramicin) and divalent (lasalocid). Worldwide, there are several anticoccidial drugs that are mixtures of low doses of synthetic anticoccidial drugs and a low dose of an ionophore.
The continuing success of this class of compounds results from a broad spectrum of activity across species and a lack of serious problems with drug resistance,10 often at a very competitive price. Chapman listed the discovery of ionophores as a milestone in avian coccidiosis research.11 McDougald added to Reid’s broad statement that anticoccidial drugs allowed the poultry industry to become more productive and efficient.12,13 Thus, the ionophores were the backbone of intensive poultry production, and without them, the industry could not have grown to the tremendous level we have today.
Initially, it was thought that ionophores would not work in the field. Heavy coccidia challenges, often used in coccidia efficacy battery studies, showed that ionophores did not have as strong anticoccidial efficacy as most synthetic anticoccidials. Even with these battery test results, the ionophores were approved and proved successful in controlling coccidiosis in the field.
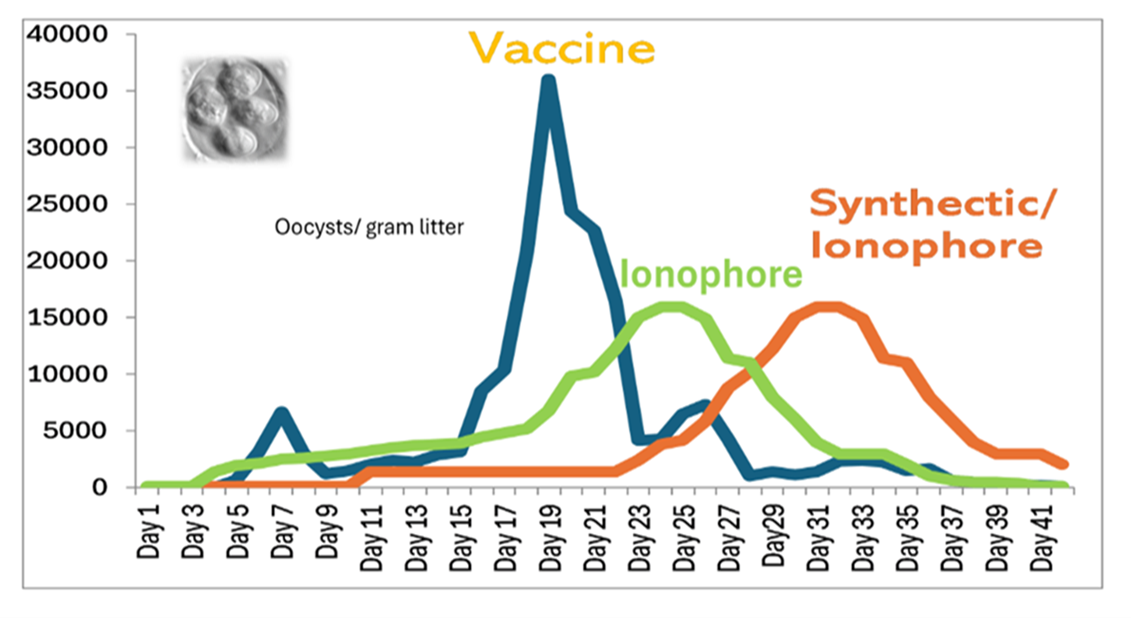
The control of field coccidiosis with ionophores was determined to be direct action against the coccidia and immunological action. The ionophores manage coccidiosis by not being fully coccidiocidal, thereby allowing some coccidia to cycle and, subsequently, coccidia immunity to develop. In this representation of a typical epidemiological oocyst-shedding pattern for vaccination and for ionophore feeding (Figure 1), oocyst shedding increases during the growout phase, with immunity developing simultaneously with each reinfection. With both programs, the number of oocysts rises and then decreases as immunity develops.
Figure 1. Oocysts shedding pattern
By not having full coccidiocidal activity, ionophores have the advantage of slower selection for drug resistance compared to many synthetic anticoccidials. Conway and McKenzie suggested that the immunological control associated with ionophore use is similar against strains that are sensitive as well as those with reduced anticoccidial sensitivity.14
As shown, all the anticoccidials have been used for many years. However, many of these drugs still have effective anticoccidial activity. Anticoccidial-sensitivity testing prior to use has been very beneficial and has contributed to their longevity.
With the successful and resilient use of the ionophorous anticoccidials, these older synthetics are often used in rotation and shuttle programs. This has reduced the duration of usage and “rested” these drugs that are more prone to resistance development. Without the use of ionophores, many of the synthetic anticoccidial drugs would not be as effective today.
Coccidiosis vaccination: Bio-shuttle
Coccidiosis vaccination has increased worldwide due to many factors, including increased incidence of drug resistance and the removal of growth-promoting feed additives. Coccidia vaccines are comprised of live, commonly occurring Eimeria species. For immunity to develop, coccidia must cycle through the birds. With each cycle, immunity is strengthened.
However, there are downsides to vaccination. Vaccination administers a live parasite, which, by its nature, will cause damage to the intestinal tract. Related to this damage is the potential for Clostridium perfringens proliferation, which can lead to necrotic enteritis. Additionally, administration issues, environment, chick quality, brooding time and many more production issues can influence cycling and uniform immunity development.
An option used by many poultry producers to manage coccidia following vaccination is a bio-shuttle or hybrid program. A bio-shuttle program is the feeding of a low-use level of a drug during the peak of vaccine-related coccidia cycling. Figure 2 illustrates that the idea is to reduce the level of infection after adequate coccidial immunity has developed, thus reducing coccidiosis damage and potentially lowering the incidence of necrotic enteritis.
With the bio-shuttle program, the drug is not intended to completely eliminate the coccidia but to reduce the coccidiosis level. As previously mentioned, the ionophores are well-suited for this type of program with a lower-use level available. Ionophores allow some coccidia to cycle, enabling coccidia immunity to develop and providing an added bonus of antimicrobial activity.
Figure 2. Salinomycin/lasalocid bio-shuttle
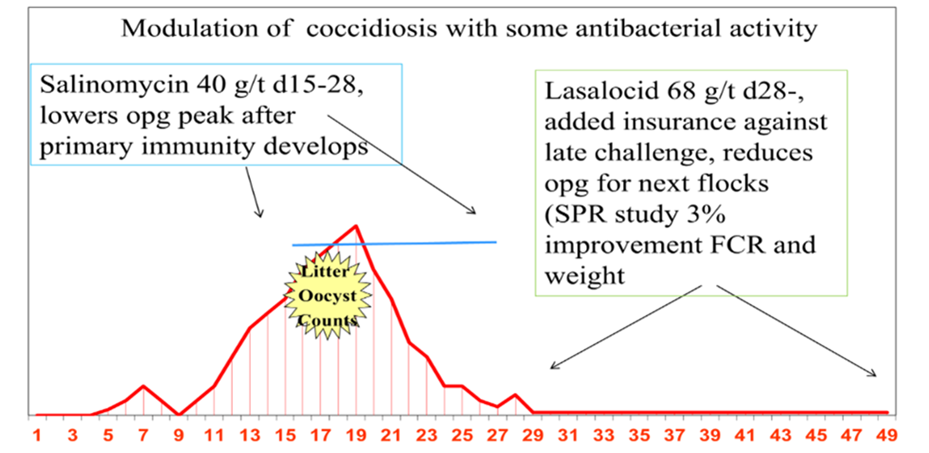
Bio-shuttle program: Low dose ionophore with coccidia vaccination. Vaccine applied at hatch. Salinomycin 40 g/t D18-28 and lasalocid 68 g/t D28-42. Mathis et al, 2017.
Conclusion
The use of ionophores as standalone, straight ionophore programs, in rotation and shuttle programs (to increase modes of action and “rest” other anticoccidials), and in bio-shuttle programs (vaccine + anticoccidial) has made this class of anticoccidial indispensable for coccidiosis control. The polyether ionophorous antibiotic anticoccidials are truly the backbone of intensive poultry production.
References
- Levine PP. Chemotherapy in the control of avian coccidiosis. In: Proceedings of the US Livestock Sanitary Association. 1941;45:118-120.
- Chapman HD. Milestones in avian coccidiosis research: A review. Poult Sci.2014;93(3):501-511.
- Grumbles LC, Delaplane JP,Higgins Sulfaquinoxaline in the control of Eimeria tenella and Eimeria necatrix in chickens on a commercial broiler farm. Science. 1948;107:196.
- Reid WM. Use of anticoccidials buy the poultry industry in the USA. Presented: Symposium International sur les Coccidioses, Tours, France. 1973;17.
- McDougald LR. Chemotherapy of Coccidiosis In: The Biology of the Coccidia. Edited by: Long Baltimore, MD: University Park Press. 1982;373-428.
- Ibid.
- Reid WM, McDougald L.R. In: 9th edition Diseases of Poultry; Protozoa/Coccidiosis. 1981;791.
- Chapman HD.Anticoccidial drug resistance. In: The Biology of the Coccidia. Edited by: Long, L.. Baltimore, MD: University Park Press. 1982; 429-445273-428.
- Bafundo KW, Cervantes HM, Mathis GF. Sensitivity of Eimeria Field isolates in the United States: Responses of Nicarbazin-containing Anticoccidials. Poult Sci. 2008;87(9):1760-1767.
- Jeffers TK. Eimeria tenella: sensitivity of recent field isolants to monensin. Avian Dis. 1978 Jan-Mar;22(1):157-61.
- Chapman HD. Milestones in avian coccidiosis research
- Reid WM. Use of anticoccidials buy the poultry industry in the USA
- McDougald LR. Chemotherapy of Coccidiosis
- Conway DP, Mckenzie ME. Poultry Coccidiosis: Diagnostic and Testing Procedures. 3rd, Ames, Iowa: Blackwell Publishing. 2007;1-168.
Editor’s note: The views expressed in this article are solely those of the authors.